Selected Publications
DNA origami-assembled metal–semiconductor junctions have been formed as a step toward application of these nanomaterials in nanoelectronics. Previously, techniques such as electroless plating, electrochemical deposition, or photochemical reduction have been used to connect metal and semiconductor nanomaterials into desired patterns on DNA templates. To improve over prior work and provide a more general framework for the creation of electronic nanodevices as an alternative nanofabrication step, we have developed a method to connect gold (Au) and tellurium (Te) nanorods on a single DNA origami template without electroplating by annealing after coating with a heat-resistant polymer. Bar DNA origami templates (17 nm × 410 nm) were seeded site-specifically with Au and Te nanorods in an alternating manner. These templates were then coated with a polymer and annealed at different temperatures. At 170 °C, the Au and Te nanorods were best connected, and we hypothesize that the junctions were established primarily due to the atomic mobility of gold. Electrical characterization of these Au/Te/Au assemblies revealed some nonlinear current–voltage curves, as well as linear plots that are explained. This annealing method and the metal–semiconductor nanomaterials that are formed simply through controlled seeding and annealing on DNA origami templates have potential to yield complex nanoelectronic devices in the future.
Several electrical devices are formed by growing vertically aligned carbon nanotube (CNT) structures directly on a substrate. In order to attain high aspect ratio CNT forest growths, a support layer for the CNT catalyst, usually alumina, is generally required. In many cases, it has been found that current can pass from a conductive substrate, across the alumina support layer, and through the CNTs with minimal resistance. This is surprising in the cases where alumina is used because alumina has a resistivity of 𝜌>1014ρ>1014 Ω cm. This paper explores the mechanism responsible for current being able to cross the alumina support layer with minimal resistance following CNT growth by using scanning transmission electron microscopy imaging, energy dispersive x-ray spectroscopy, secondary ion mass spectroscopy, and two-point current-voltage (I-V) measurements. Through these methods, it is determined that exposure to the carbonaceous gas used during the CNT growth process is primarily responsible for this phenomenon.
Bottom-up fabrication using DNA is a promising approach for the creation of nanoarchitectures. Accordingly, nanomaterials with specific electronic, photonic, or other functions are precisely and programmably positioned on DNA nanostructures from a disordered collection of smaller parts. These self-assembled structures offer significant potential in many domains such as sensing, drug delivery, and electronic device manufacturing. This review describes recent progress in organizing nanoscale morphologies of metals, semiconductors, and carbon nanotubes using DNA templates. We describe common substrates, DNA templates, seeding, plating, nanomaterial placement, and methods for structural and electrical characterization. Finally, our outlook for DNA-enabled bottom-up nanofabrication of materials is presented.
Self-assembly nanofabrication is increasingly appealing in complex nanostructures, as it requires fewer materials and has potential to reduce feature sizes. The use of DNA to control nanoscale and microscale features is promising but not fully developed. In this work, we study self-assembled DNA nanotubes to fabricate gold nanowires for use as interconnects in future nanoelectronic devices. We evaluate two approaches for seeding, gold and palladium, both using gold electroless plating to connect the seeds. These gold nanowires are characterized electrically utilizing electron beam induced deposition of tungsten and four-point probe techniques. Measured resistivity values for 15 successfully studied wires are between 9.3 × 10−6 and 1.2 × 10−3 Ωm. Our work yields new insights into reproducible formation and characterization of metal nanowires on DNA nanotubes, making them promising templates for future nanowires in complex electronic circuitry.
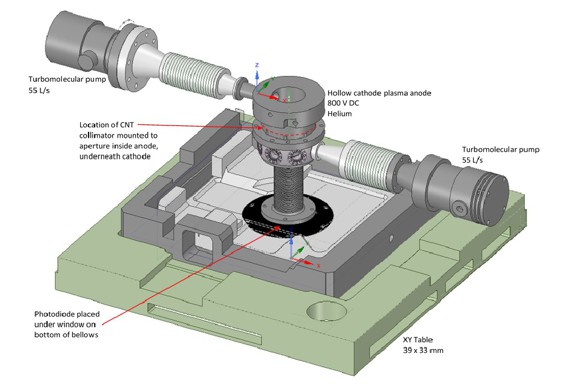
We report on a large-area, high-aspect-ratio, carbon nanotube (CNT) forest structure produced at BYU acting as a window/separator for a hollow cathode EUV lamp. The structure has large-surface-area, high light trans-mission, and differential pumping. CNT fabrication allows for variable dimensions, which allows various EUV distributions and pressure gradients to be possible. Theory is presented for predicting such distributions and gradients. Several structures have been fabricated; their dimensions, properties, and predicted distributions and gradients are given.
DNA origami templated fabrication enables bottom-up fabrication of nanoscale structures from a variety of functional materials, including metal nanowires. We studied the impact of low temperature annealing on the morphology and conductance of DNA templated nanowires. Nanowires were formed by selective seeding of gold nanorods on DNA origami and gold electroless plating of the seeded structures. At low annealing temperatures (160° C for seeded-only and 180° C for plated) the wires broke up and separated into multiple, isolated islands. Through the use of polymer-constrained annealing, the island formation in plated wires was suppressed up to annealing temperatures of 210° C. Four-point electrical measurements showed that wires remained conductive after a polymer-constrained anneal at 200° C.